Blackpoll Warbler Setophaga striata Scientific name definitions
- NT Near Threatened
- Names (44)
- Monotypic
William DeLuca, Rebecca Holberton, Pamela D. Hunt, and Bonita C. Eliason
Version: 1.0 — Published March 4, 2020
Text last updated June 4, 2013
Text last updated June 4, 2013
Sign in to see your badges
Species names in all available languages
| Language | Common name |
|---|---|
| Catalan | bosquerola estriada |
| Czech | lesňáček černohlavý |
| Danish | Stribet Sanger |
| Dutch | Zwartkopzanger |
| English | Blackpoll Warbler |
| English (United States) | Blackpoll Warbler |
| Finnish | viirukerttuli |
| French | Paruline rayée |
| French (France) | Paruline rayée |
| German | Streifenwaldsänger |
| Greek | Μελανοπάρουλα |
| Haitian Creole (Haiti) | Ti Tchit sèjan |
| Hebrew | סבכון פסים |
| Hungarian | Kucsmás lombjáró |
| Icelandic | Rákaskríkja |
| Japanese | ズグロアメリカムシクイ |
| Lithuanian | Dryžuotasis krūminukas |
| Norwegian | svarthetteparula |
| Polish | lasówka czarnogłowa |
| Portuguese (Brazil) | mariquita-de-perna-clara |
| Portuguese (Portugal) | Mariquita-de-perna-clara |
| Romanian | Frunzar striat |
| Russian | Пестрогрудая древесница |
| Serbian | Prugasta cvrkutarka |
| Slovak | horárik tajgový |
| Slovenian | Črnoglavi gozdičar |
| Spanish | Reinita Estriada |
| Spanish (Argentina) | Arañero Estriado |
| Spanish (Chile) | Monjita americana |
| Spanish (Costa Rica) | Reinita Rayada |
| Spanish (Cuba) | Bijirita de cabeza negra |
| Spanish (Dominican Republic) | Cigüita Cabeza Negra |
| Spanish (Ecuador) | Reinita Estriada |
| Spanish (Honduras) | Chipe Copa Negra |
| Spanish (Mexico) | Chipe Cabeza Negra |
| Spanish (Panama) | Reinita Estriada |
| Spanish (Peru) | Reinita Estriada |
| Spanish (Puerto Rico) | Reinita Rayada |
| Spanish (Spain) | Reinita estriada |
| Spanish (Uruguay) | Arañero Estriado |
| Spanish (Venezuela) | Reinita Rayada |
| Swedish | vitkindad skogssångare |
| Turkish | Kara Kırçıllı Ötleğen |
| Ukrainian | Пісняр-лісовик білощокий |
Setophaga striata (Forster, 1772)
PROTONYM:
Muscicapa striata
Forster, 1772. Philosophical Transactions 62, p.428.
TYPE LOCALITY:
Severn River [= Fort Severn, west coast of Hudson Bay, fide Amer. Ornith. Union, 1910, Checkhst North Amer. Birds, ed. 3, p. 315].
SOURCE:
Avibase, 2023
Definitions
- SETOPHAGA
- striata
The Key to Scientific Names
Legend Overview
UPPERCASE: current genus
Uppercase first letter: generic synonym
● and ● See: generic homonyms
lowercase: species and subspecies
●: early names, variants, misspellings
‡: extinct
†: type species
Gr.: ancient Greek
L.: Latin
<: derived from
syn: synonym of
/: separates historical and modern geographic names
ex: based on
TL: type locality
OD: original diagnosis (genus) or original description (species)
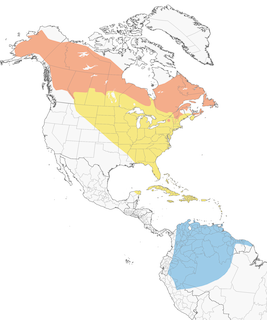
- Year-round
- Migration
- Breeding
- Non-Breeding
Distribution of the Blackpoll Warbler





























