Horned Curassow Pauxi unicornis Scientific name definitions
- CR Critically Endangered
- Names (19)
- Monotypic
Josep del Hoyo, Guy M. Kirwan, and David Christie
Version: 1.0 — Published March 4, 2020
Text last updated September 17, 2015
Text last updated September 17, 2015
Sign in to see your badges
Species names in all available languages
| Language | Common name |
|---|---|
| Catalan | hoco unicorne |
| Czech | hoko bolívijský |
| Dutch | Hoornhokko |
| English | Horned Curassow |
| English (United States) | Horned Curassow |
| French | Hocco unicorne |
| French (France) | Hocco unicorne |
| German | Hornhokko |
| Japanese | ミナミカブトホウカンチョウ |
| Norwegian | sørhjelmhokko |
| Polish | czubacz rogaty |
| Russian | Рогатый гокко |
| Serbian | Rogati hoko |
| Slovak | hoko rohatý |
| Spanish | Paují Unicornio Boliviano |
| Spanish (Spain) | Paují unicornio boliviano |
| Swedish | sydlig hjälmhocko |
| Turkish | Bolivya Boynuzlu Hokkosu |
| Ukrainian | Кракс-рогань південний |
Pauxi unicornis Bond & Meyer de Schauensee, 1939
PROTONYM:
Pauxi unicornis
Bond & Meyer de Schauensee, 1939. Notulae Naturae no.29, p.1.
TYPE LOCALITY:
SOURCE:
Avibase, 2023
Definitions
- PAUXI
- pauxi
- unicornis
The Key to Scientific Names
Legend Overview
UPPERCASE: current genus
Uppercase first letter: generic synonym
● and ● See: generic homonyms
lowercase: species and subspecies
●: early names, variants, misspellings
‡: extinct
†: type species
Gr.: ancient Greek
L.: Latin
<: derived from
syn: synonym of
/: separates historical and modern geographic names
ex: based on
TL: type locality
OD: original diagnosis (genus) or original description (species)
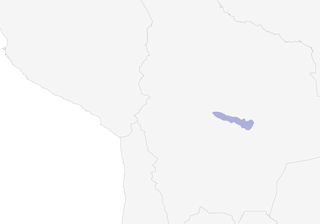
- Year-round
- Migration
- Breeding
- Non-Breeding
Distribution of the Horned Curassow
























